|
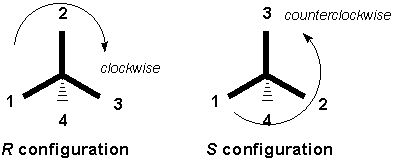
2) Rotate the other bond in the plane until the 4 priority group is in a position (dashed or wedged) from which you can more easily assign R and S. The basis is again the CIP priority rules. There are 3 ways to approach correctly assigning R and S in such a scenario: 1) Try to visualize the molecule in your mind from the correct perspective. In the case of a double bond, the traditional cis/ trans nomenclature system is not sufficiently accurate and the E-/ Z- is currently preferred. The R-/S- notation is valid only for the absolute configuration of a center having single bonds only. On the other hand, S means left by the Latin, ‘sinister’ refers to left. 
(4)More atomic number containing atom and how many atoms are present of the higher atomic number containing elements. Is clockwise rotation R, or S configuration R meaning right by the Latin word ‘rectus’ refers to the right side rotation of plane-polarized light. (3)If the first atom of two same groups have same substitutes of higher no will get more priority. (2)When two or more groups have similar first atom, the priority is determined by considering the atomic number of second atom. (1)According to the atomic number when only atom is present and in a group the direct attached atom is considered with atomic number. If it is going clockwise, then it is an R-enantiomer If it is going counterclockwise, it is an S-enantiomer. Follow the direction of the remaining 3 priorities from highest to lowest priority (lowest to highest number, 1<2<3).Ī counterclockwise direction is an S ( sinister, Latin for left) configuration.Ī clockwise direction is an R ( rectus, Latin for right) configuration.ĭirection of the travel 1-2-3 dictates configuration Second, point the lowest priority (4) atom away from you. R- and S-notation use the CIP priority rules for the assignment of the absolute configuration around a stereocenter.įirst, assign priorities as described above to each bonded group surrounding the stereocenter (1, highest to 4, lowest). Realize that when you do this it will mean that sometimes groups with higher total weights will have lower priority because of a lower weight of the atom that connects them. If this does not assign priority, consider the next atoms until there is a difference. Place the 4 molecule in the back of the chiral center and then in a clockwise (R) or counterclockwise (S) direction label the bonds with atoms 1, 2, 3. If the first atom of two groups is the same, consider the second atom(s) in the same way as the first.An atom with higher atomic number has higher priority. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
